Plasma concentrations of ARI, DARI and OLA were quantified by a high‐performance liquid chromatography–tandem mass spectrometry method developed in our laboratory. Subsequently, the samples were centrifuged at 1900 g for 10 minutes and then the plasma was collected and stored at −20☌ until the determination of drug concentrations. Each blood sample was labelled with the protocol code, volunteer number, treatment period and day and extraction time without specifying the administered drug. Twenty‐two blood samples were collected from each participant for pharmacokinetic assessments during each period, thus 44 samples in total: 7 samples on day 1 (predose and 1, 2, 3, 5, 8 and 12 hours after dosing) 1 (predose) sample on days 2, 3 and 4 7 samples on day 5 (predose and 1, 2, 3, 5, 8 and 12 hours after dosing) and 1 sample on days 6, 7, 9, 11 and 15 (corresponding to 24, 48, 96, 144 and 240 hours after the last dose, respectively). The random allocation sequence, the recruitment of participants and their assignment to interventions were performed by investigators of the Clinical Trials Unit. In the second period, after a washout period of 28 days, each volunteer received the opposite drug they received in the first period. 
The subjects were hospitalized from 1 hour before the first dose until 24 hours after the last dose. 19 The drug was administered at 09:00 each day under fasting conditions. 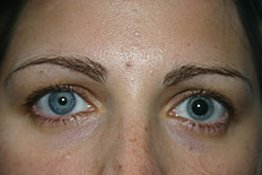
Block randomization was used to assign the treatment to each volunteer on the first day. ARI 10 mg/day tablets or OLA 5 mg/day film‐coated tablets were administered during 5 consecutive days. The clinical trial was phase I with multiple oral dose design, open‐label, randomized, crossover, 2‐periods, 2‐sequences, single‐centre and comparative study performed between June 2018–April 2019. Furthermore, their relationship with pharmacokinetics and pharmacogenetics was also evaluated. The aim of the current study was to evaluate if ARI and OLA affect pupillometric parameters in healthy subjects after multiple dose administration. 17 Accordingly, genetic polymorphisms present in these genes can affect pupil response. 14 By contrast, these drugs could affect the pupil diameter due to their affinity for dopamine and serotonin receptors 15, 16 as serotonin and dopamine are effectors on various types of muscles including the sphincter pupillae and the dilator pupillae. It can be due to inducing unopposed parasympathetic stimulation of the pupil with significant α1‐adrenergic receptor blockade. Several atypical antipsychotics caused pupil miosis in overdose patients. 9 It was thoroughly described that opioid drugs cause pupil constriction (miosis). Pupillography is a noninvasive and cost‐effective method to determine autonomic nerve activity, 8 which was developed in 1958. 5 Olanzapine is predominantly metabolized by direct glucuronidation via the UDP‐glucuronosyltransferase (UGT) enzyme family, principally by UGT1A4, 6 CYP1A2 and to a lesser extent by CYP2D6 and CYP3A4. Moreover, the pharmacological activity of DARI is similar to ARI. Dehydro‐aripiprazole (DARI), its main active metabolite, accounts for 40% of the parent compound in plasma. 3, 4ĪRI is extensively metabolized by cytochrome P450 (CYP) enzymes CYP3A4 and CYP2D6. 
Additionally, it has antagonistic activity at dopamine D3 and D4, serotonin 5‐HT3 and 5‐HT6, histamine H1, α1‐adrenergic, and muscarinic M1–5 receptors. 2 OLA has higher antagonistic affinity for 5‐HT2A serotonin receptors than for D2 dopamine receptors. 1 ARI has partial agonistic activity at dopamine D2, D3, D4 and serotonin 5‐HT1A, 5‐HT2C as well as α1‐adrenergic receptors and also exhibits 5‐HT2A and 5‐HT7 receptor antagonism. Aripiprazole (ARI) and olanzapine (OLA) are atypical (second generation) antipsychotics commonly prescribed for patients with schizophrenia or schizoaffective disorders. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
